A vaccine being tested in human patients by Dublin-based company United Neurosciences has shown promising results, according to a statement released by the company.
A number of biotechs have aimed to resolve Alzheimer’s via a vaccine over recent years, only to call it off due to nasty side effects like brain swelling. But this drug, which the company has dubbed UB-311, is offering hope after wrapping up a Phase 2a Clinical Study. Next, the drug will begin a 2b study, which will further determine efficacy, before going on to a Phase 3 study if all is successful. Phase 3 is the necessary trial before drug approval and is tested on large groups of patients.
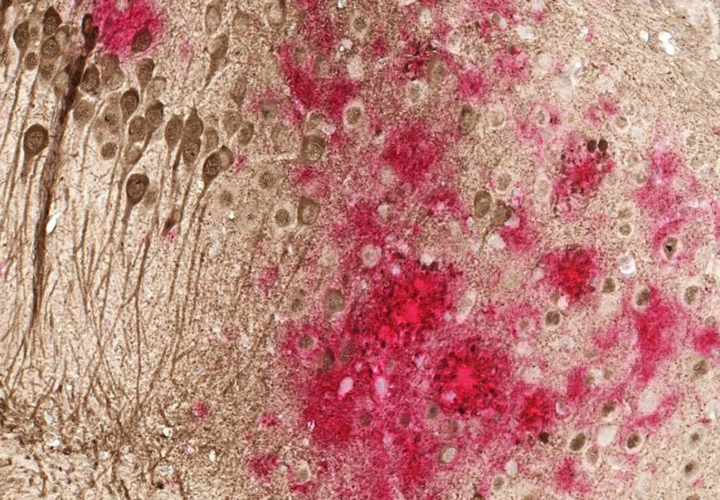
96 percent of patients responded to the drug and showed improved cognition and a lower level of beta-amyloid, the toxic plaque that accumulates in Alzheimer’s patients and the target of the vaccine.
For the Phase 2a study, the drug was tested amongst a group of 42 patients with mild cognitive impairment who were likely in the early stages of Alzheimer’s. The group was divided in three; one was given a placebo and the other two were given the vaccine, administered via shots, three times and then followed up with booster shots either every three or six months.
Of the patients, 96 percent responded to the drug and showed improved cognition and a lower level of beta-amyloid, the toxic plaque that accumulates in Alzheimer’s patients and the target of the vaccine.
“The positive results show that we can safely raise and maintain anti-[beta-amyloid] antibody titers in a predictable and sustained manner,” said Peter Powchik, executive vice president of research and development at United Neurosciences. “High response rates, reproducibility of response and generation of antibodies directed to relevant toxic protein species are key elements of an effective therapeutic vaccine for neurodegenerative conditions. The UNS platform is proving that it can deliver on these requirements.”
And while the results are encouraging enough for United Neurosciences to continue pursuing the vaccine with the same patients, the small number of patients tested means the results are not statistically significant. Additional results will likely be announced in March at the 14th International Conference on Alzheimer’s and Parkinson’s Diseases in Lisbon, Portugal.
“These early results suggest a clinical response and support the continued and rapid development of UB-311. The intent of this Phase 2a study was to acquire directional information on the safety, tolerability and therapeutic potential of UB-311 in patients with Alzheimer’s disease,” said Chief Executive Officer Mei Mei Hu.
A similar vaccine was tested with success on mice by scientists at the University of Texas Southwestern Medical Center and announced in November 2018. Both that vaccine and UB-311 operate under the assumption that beta-amyloid is the cause of the debilitating symptoms of Alzheimer’s, though it’s not an assumption everyone agrees with. In fact, other clinical trials targeting beta-amyloid have recently been called off by drug companies, calling the origin of Alzheimer’s into question.
Regardless, both vaccines would require that older adults get access to early diagnostic tools that could determine who is most at risk for Alzheimer’s and would benefit from a vaccine.




How does this compare to the Biogen studies in the US and what is being done for short term memory problems?
I would like to get this shot how can I?
Yes, how can you get this medicine.
My mothers family as well as herself has passed from altimeters/dementia. i myself would love to be a trial candidate for testing this. I feel i could very well be assessable to this disease. as a matter of fact, i know i am. i am 59 years old and have researched my entire life into this horrible disease and have watched so many loved ones die from this. As well as being a helper to families that needed that extra hand in helping sit and caring for their loved one. May i please have the privaledge in being part of bringing this horrible disease to a hault!
Don’t be a guinea pig… there are plenty of things you can do to prevent alz… start taking pure virgin organic coconut oil,,, it’s everywhere but costly in most places… costco’s is an extremely reasonable price… use it in your coffee. Mix it with your butter 3pt coco 1 butter. start using Moringa leaf powder… good quality stuff. Listen to binaural beats for syncing your hemispheres in the noggin. Turmeric with piperine… organic high quality… mix it with good honey… yummm. Tons of research on the net of other things to do… oh and don’t forget exercise !
bj…..
Your comment is so ignorant. If it were only that simple. I have seen 6 family members (all female) die from this horrible disease. All were extremely health conscious. Stop blaming the victims.
There are other studies in the US and world-wide for healthy elderly who have genetic markers but no cognitive impairment. This is a worldwide study by Amgen and Novartis called Generation One and Generation 2. Lots of exciting things happening to prevent AD.
That’s really good news for us. Lets have hope that UB-311 will come into play with more effectiveness in curing the Alzheimer disease.
I need this.
I read the article relating to the UB-311 drug and I would really like to enroll in the Phase 3 Clinical Study. Hopefully you can accept into this test/study. Thanks and I hope to hear back from you with a positive response. Best regards. Tom
My mother and father has Alzheimer’s.it is just awful.my mother has parkinsins
I would be interested in this ! Please let me know more !!
Please let me know how I can enter the Phase 3 clinical trail for the UB311 drug/vaccine.
Please let me know who I can contact and a telephone number to get this?
My father has the early onset of Alzheimer’s and has been tackling himself in various ways including the use of coconut oil and MCT oil. Is there any chance that this vaccine could be available to him?
How long is Phase 3, and when would it begin?
I would like to also join the study.
My sister was diagnosed 3 years ago at age 64
I would like to get her into this trial for the vaccine
I have a friend with advanced Alzheimer’s. I hope she would be a candidate for the study.
Can this be beneficial for those who have early Alzheimer’s disease?
PLEASE LET ME BECOME PART OF THIS RESEARCH. I AM A 59 YEARS OLD FEMALE CAUCASIAN AND HAVE LOST SO MANY OF MY FAMILY MEMBERS AS WELL AS MANY LOVED ONES TO THIS HORRIBLE DISEASE. IVE ALSO BEEN TOLD I SHOW SIGNS OF EARLY ONSET MYSELF, BUT DONT KNOW WHAT TO DO ABOUT IT. I DO NOT WANNA DIE AS I HAVE WATCHED SO MANY DO BECAUSE OF THIS MONSTER! I LIVE IN THE ARK-LA-TEX AREA AND DALLAS IS NOT THAT FAR AWAY. MAY I PLEASE BE PART OF THIS RESEARCH AND MAY I BE A CANDIDATE FOR THIS PROMISING EXPERIMENT? I HAVE TOOK HUPERZINE AS WELL AS MANY OTHER MINERALS DURING MY LIFETIME FOR HOPEFUL PREVENTION.
This is called the Universial Vaccine
Is this available now to get? I would be very much interested in finding out how to get this.
I would like my husband to be part of this trial. He has MCI? It seems to be his spacial energy that is affected. When he goes to go right he goes left. His memory is not that bad and he does not repeat himself. I have researched all over the world for to make his life as good as I can. Thank you Anne Morrison
How can one be enrolled in Phase3 trials?
Hi my husband 36 has the psn1 gene, very interested in doing all we can to prevent eoad developing. We haven’t had a diagnosis tet – how can we get involved?
I would like to enroll in this treatment for Alzheimer’s. Please let me know how to enroll. Thanks. Tom
I am willing to be a trial patient.
I’d love to take part in this trial
PLEASE ADVISE HOW TO GET INTO THE CLINICAL TRIAL.
Mu husband has just been diagnosed. How can he participate in your study? He will be 64 in November, is a non smoker, non drinker, and has never done recreational drugs during our marriage of 37 years. He is taking his first dose of Namenda starter pack tonight. There may be a genetic link, but this has not been confirmed.
Alzheimer’s diagnosis has become a reality recently with my husband and am willing to go the extra mile, and do what needs to be done to give him the chance of a quality of life that he has given me. How do I get him in this trial? Who do I need to speak to? What do I need to do? Please respond, please be an answer to my prayers. I will travel anywhere, And not be a burden to anyone. I will support fully all of your criteria.