TAUVID has been FDA-approved to image tau neurofibrillary tangles in people's brains as a means of diagnosing Alzheimer's.
Scanning technology is the most definitive way of diagnosing Alzheimer’s disease. One of the most accurate is called a positron emission tomography (PET) scan, and has been able to detect beta amyloid plaque, the substance considered the early marker of Alzheimer’s disease. Now, a new imaging drug used with a PET scan has been approved by the FDA to target tau protein tangles, another hallmark of Alzheimer’s disease, revealing the tangled substances that disrupt the neuron transport system in the brain.
TAUVID, a drug manufactured by Avid Radiopharmaceuticals, is being used on patients with cognitive impairment who are being evaluated for Alzheimer’s.
Charles Ganley, MD, director of the Office of Specialty Medicine in the FDA’s Center for Drug Evaluation and Research, said in a statement that “while there are FDA approved imaging drugs for amyloid pathology, this is the first drug approved for imaging tau pathology, 1 of the 2 neuropathological hallmarks of Alzheimer’s disease, and represents a major advance for patients with cognitive impairment being evaluated for the condition.”
Avid Radiopharmaceuticals, a daughter company of Eli Lilly and Company (Lilly), has previously developed AMYVID to image beta-amyloid plaques in Alzheimer’s patients. Mark Mintun, MD, vice president of Lilly’s Pain and Neurodegeneration Research and Development, said that he is “excited that TAUVID has now been approved to image tau neurofibrillary tangles, which is the other key pathology, allowing a more comprehensive evaluation of patients.”
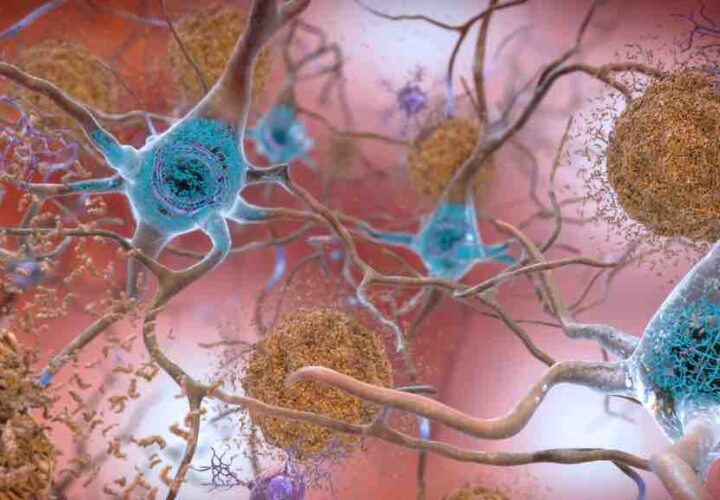
TAUVID acts as an injected tracer or dye and binds to the regions in the brain associated with tau protein tangles, and can be visualized through a PET scan.
Prior to the release of the FDA-approved TAUVID, postmortem brain examinations were the only way to identify and observe tau tangles in the brain.
PET scans are not commonly used in medical practices because of the high expense and are often not reimbursable by insurance plans. A scan can typically cost five to seven thousand dollars.
Innovation in cheaper diagnostics is advancing with companies moving closer to producing blood tests to detect biomarkers in the blood of people with Alzheimer’s. Eye scans and skin biopsies are also being studied as ways to detect early signs of Alzheimer’s disease.
In the initial stages, TAUVID will be limited in availability. Further commercial demand and payor reimbursement will influence its expansion.
UPDATE, 20 November 2023: As of a landmark policy change in October 2023, Medicare now covers beta-amyloid PET scans for diagnosing Alzheimer’s disease.