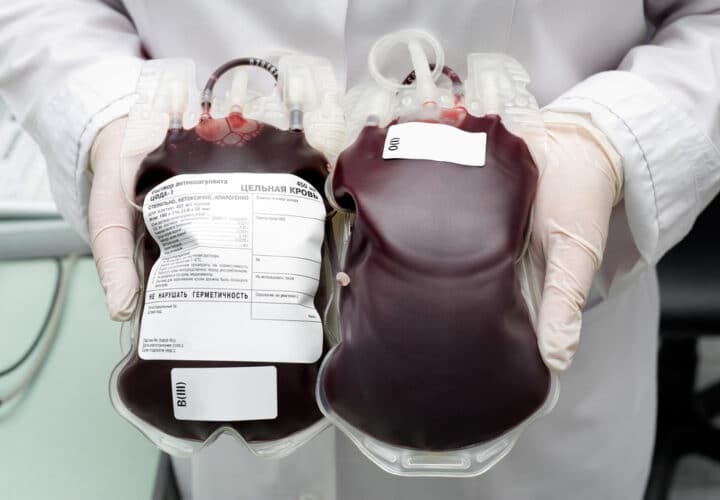
It sounds like something out of a science fiction movie with a Goop-esque twist: a company called Ambrosia offers plasma transfusions of “young” blood into older people, hoping that the process would help halt the aging process. But not anymore: On Tuesday, the F.D.A. released a warning in a statement stating that there is “no proven clinical benefit of infusion of plasma from young donors to cure, mitigate, treat, or prevent” conditions like aging or Alzheimer’s. Ambrosia released a statement on their website that they would be ceasing treatments.
The idea of using the plasma of young people to slow aging gained popularity back in 2017 when a very small study conducted by Stanford University found that people with Alzheimer’s who were given transfusions of young plasma performed slightly better in everyday tasks like dressing themselves.
That study was based on previous findings that 20-month-old mice (the human equivalent of 65-year-olds) who were given the plasma of three-month-old mice (the human equivalent of 20-year-olds) did better on a water maze.
The two studies were not affiliated with Ambrosia, whose founder Jesse Karmazin is a Stanford medical school graduate but not a licensed practicing professional. Prior to Tuesday, Ambrosia was charging $8,000 per one liter of young blood, or $12,000 for two liters in Los Angeles; San Francisco; Tampa, Florida; Omaha, Nebraska; and Houston, Texas.
Researchers who have worked on plasma transfusions in mice called Karmazin’s process “dangerous,” according to Business Insider. The company did conduct a clinical trial and charged participants $8,000 to participate, but those results have not been made public.
The F.D.A. made clear that while clinical trials like Stanford’s study on humans and mice will be allowed to continue, but the plasma offered at clinics has not been proven to be safe or effective.
“Simply put, we’re concerned that some patients are being preyed upon by unscrupulous actors touting treatments of plasma from young donors as cures and remedies. Such treatments have no proven clinical benefits for the uses for which these clinics are advertising them and are potentially harmful,” said Scott Gottlieb, M.D., F.D.A. Commissioner and Peter Mark, M.D., Ph.D., a director at the F.D.A.’s Center for Biologics Evaluation and Research in a joint statement.
Researchers who have worked on large-scale clinical trials using young blood plasma agree.
“There’s just no clinical evidence [that the treatment will be beneficial], and you’re basically abusing people’s trust and the public excitement around this,” said neuroscientist Tony Wyss-Coray of Stanford University to Science.
The F.D.A. recommends that people who want treatments to ask their health care providers if the F.D.A. has reviewed the treatment. “You also can ask the clinical investigator to give you the F.D.A.-issued I.N.D. number and to provide a copy of the F.D.A. communication acknowledging the I.N.D.,” said Gottlieb and Mark. Consumers should report adverse effects to the MedWatch Reporting Program.
Common risks of plasma infusions, even for situations that they’re recommended for, include allergic reactions and transfusion associated circulatory overload. Less common risks include transfusion related acute lung injury or transfusion associated circulatory overload and infectious disease transmission, according to the F.D.A.