Clinical trials are recruiting younger, healthier participants for Alzheimer's studies like the new TRAILBLAZER 3 donanemab clinical trial.
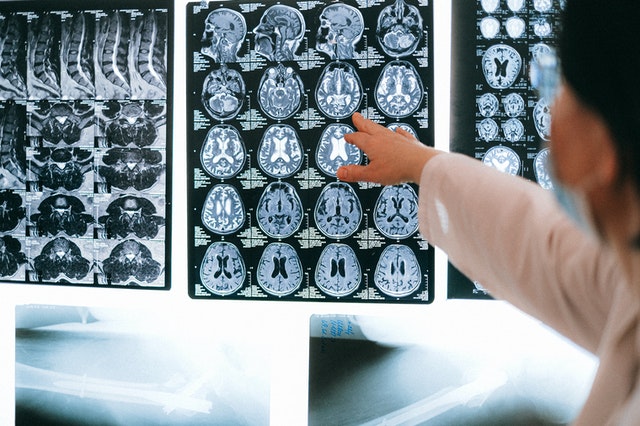
Alzheimer’s is a notoriously difficult disease to identify with certainty. By the time a patient receives a diagnosis, its pathology in the brain is already widespread. This has been a major setback in developing treatments that can slow or stop its progression, as mounting evidence indicates that the optimal time for treating the disease may be before its telltale symptoms are even detectable.
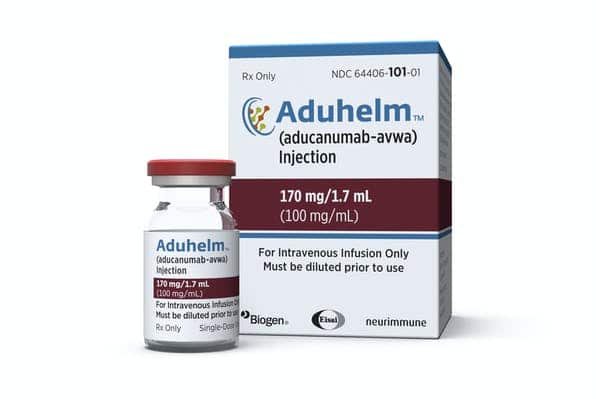
Biogen’s Aduhelm treatment, which was approved by the Food and Drug Administration earlier this year, was a major milestone in that, even amid roiling controversy, it became the first FDA-approved treatment for Alzheimer’s that is designed to treat the underlying pathology of the disease, rather than only addressing its symptoms at a surface level. While its efficacy has been questioned and its approval has been called premature, its approval has helped pave the way for similar drugs: Eli Lilly’s anti-amyloid donanemab is not far behind in the FDA pipeline.
This summer, donanemab received the FDA’s breakthrough therapy designation, and the results of its TRAILBLAZER 2 clinical trials completed this year were encouraging: Trial data did indicate the drug is effective in slowing cognitive decline in people who have already developed Alzheimer’s. But it also provided researchers with an extra clue.
According to Dr. Edgardo Rivera, a clinical trial administrator at Charter Research in Florida, TRAILBLAZER 2 data suggests that the drug may work better at earlier stages of the disease — before symptoms like memory impairment appear.
“Pursuing clinical trials in healthy people comes out of the amyloid hypothesis,” Dr. Julio C. Rojas, neurologist at the University of California San Francisco explained. “There’s accumulation of an abnormal protein called amyloid in the brain, that leads to accumulation of another protein, a second protein called tau, inside neurons.”
Since this process may start several decades before memory impairment or cognitive decline, reducing amyloid in the brain early may be key to preventing Alzheimer’s. Even if these preventative studies don’t find amyloid as the driver of Alzheimer’s, these trials could help identify better biomarkers and drive the development of better tools for assessing clinical improvement.
“I’m sure better treatments will come with better screening tools and psychological testing,” Rojas added.
Preventative trials also call for a major shift in diagnostic technology: Researchers are making great strides in developing new diagnostic tools for Alzheimer’s that can detect the disease years, or even decades, earlier than current methods. It also calls for an important shift in drug development — a new approach to clinical trial recruitment. Instead of recruiting people in the early stages of symptomatic Alzheimer’s, trial recruiters may need to focus on asymptomatic people who have biomarkers that indicate they are at risk.
Instead of treating individuals in this group who do develop Alzheimer’s by trying to slow the disease down once its symptoms begin inhibiting day-to-day life, new treatments could ideally stop the disease before it presents, and prevent symptoms from ever appearing.
Trial administrators for TRAILBLAZER 3 endeavors to do just that: recruiting cognitively and generally healthy people with biomarkers of Alzheimer’s disease, but without any clinical symptoms of cognitive decline. In particular, trial administrators will be looking for participants who have high levels of tau in the blood, which — along with beta-amyloid — drives Alzheimer’s pathology.
According to Eli Lilly, the trial will compare how well donanemab changes the trajectory of cognitive decline, compared to placebo, by using blood testing to search for tau.