Most Alzheimer’s research has focused on beta-amyloid plaques, but a new study finds tau protein tangles can better predict later brain atrophy.
Much of Alzheimer’s research has focused on beta-amyloid plaques, one of the main drivers behind neurodegeneration in people with dementia. But more and more research is focusing on other potential causes of these diseases — like inflammation and tau protein tangles.
In a new study out of the University of California San Francisco (UCSF) Memory and Aging Center, researchers honed in on tau protein in particular, and found that these tangles were able to better predict brain atrophy in Alzheimer’s patients than amyloid plaques.
Identifying tau in early Alzheimer’s detection
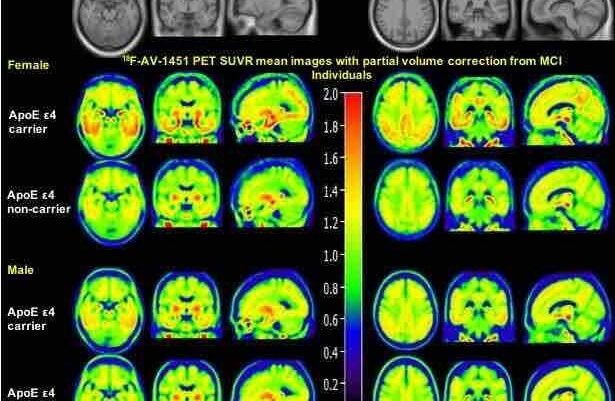
The researchers brought in 32 participants, all of whom had early clinical stage Alzheimer’s disease. They measured both amyloid and tau in their brains through PET scans, and tested brain structure at the beginning and again at the end of the study with MRIs.
They found that tau protein in particular was able to predict brain degeneration and shrinking about 15 months later. In fact, tau predicted future brain atrophy with 40 percent accuracy, while amyloid only predicted about 3 percent of brain degeneration.
The researchers concluded that tau protein may influence the development of Alzheimer’s more so than beta-amyloid, despite previous beliefs.
“The match between the spread of tau and what happened to the brain in the following year was really striking,” Dr. Gil Rabinovici, a neurologist and leader of the PET imaging program at UCSF, said in a news release.
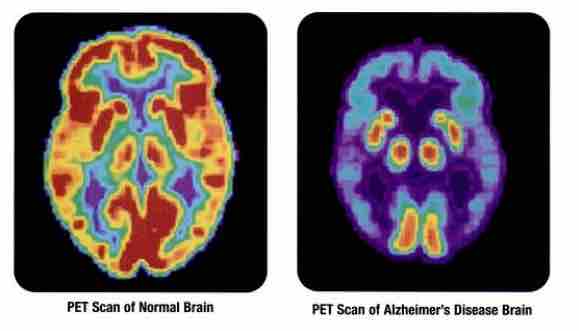
The researchers used tau-based positron emission tomography, or PET, scans to examine tau buildup in the brain. At the PET imaging program at UCSF, referred to as the “Rab Lab,” researchers use new neuroimaging techniques to identify biomarkers of dementia, to help spur earlier detection. In the past, doctors were only able to test for Alzheimer’s during autopsy.
Being able to test for beta-amyloid or tau through PET scans is helping doctors piece together important biomarkers in the brain to help diagnose dementia more quickly. Early detection is crucial in helping patients get treatment and care as soon as possible.
“Tau PET imaging predicted not only how much atrophy we would see, but also where it would happen,” Rabinovici continued. “These predictions were much more powerful than anything we’ve been able to do with other imaging tools, and add to evidence that tau is a major driver of the disease.”
Amyloid vs. tau protein in the brain
Researchers belonging to the “amyloid camp” have long believed that beta-amyloid is the main cause behind neurodegenerative diseases. But various amyloid studies and trials have been slashed in recent years, calling the amyloid theory into question.
“No one doubts that amyloid plays a role in Alzheimer’s disease, but more and more tau findings are beginning to shift how people think about what is actually driving the disease,” Renaud La Joie, a postdoctoral researcher in the In Vivo Molecular Neuroimaging Lab who also worked on the study, said in the news release.
As part of some of the latest research on tau protein, scientists have honed in on a gene known as APOE4 and its role in the development of tangles.
In a past interview with Being Patient, Jeffrey Kordower, Director of Neurobiology at Rush University, said that tau appears to have more of an effect on neurodegeneration than amyloid, possibly making it a better target for new drugs and therapies.
“We now have the ability to clear these [beta-amyloid] plaques,” Kordower said. “But when you clear these plaques nothing happens… I think that the correlation between [amyloid] plaque load and cognitive dysfunction is very poor. And the correlation of tau pathology is much better. Those connections are probably the most that correlate with cognitive dysfunction.”
The second aspect of the latest study that will hold useful for the future is the use of PET imaging to identify tau protein.
“Tau PET could be an extremely valuable precision medicine tool for future clinical trials,” Rabinovici said in the news release. “The ability to sensitively track tau accumulation in living patients would for the first time let clinical researchers seek out treatments that can slow down or even prevent the specific pattern of brain atrophy predicted for each patient.”