Researchers studied how inflammation triggers amyloid accumulation and plays a big role in the progression of brain diseases like Alzheimer's.
Scientists have largely focused on the buildup of beta-amyloid as one of the major drivers of Alzheimer’s disease, but more and more research is pointing to inflammation as another big factor.
A new study focused on brain tissue analysis concluded that inflammation may actually trigger amyloid accumulation, playing a big role in the progression of neurological degeneration and associated brain diseases.
The study, conducted by an international research team led by the German Center for Neurodegenerative Diseases (DZNE) and the University of Bonn, was recently published in the journal Nature.
For this study, the researchers examined tissue samples from the brains of deceased frontotemporal dementia (FTD) patients, cultured brain cells and mice that exhibited hallmarks of Alzheimer’s and FTD.
Tau proteins usually stabilize a neuron’s skeleton, but in degenerative brain diseases such as Alzheimer’s, the proteins are chemically altered and instead begin sticking together. This alteration of the tau proteins, and its subsequent effect on the overall stability of the brain cells, causes cell death and leads to brain disorders.
Inflammation and the Amyloid Cascade Hypothesis
The new study found that these alterations are often triggered by inflammation caused by the brain’s immune response. The researchers focused specifically on a particular protein complex, the NLRP3 inflammasome, a macromolecule located in the brain’s immune cells.
“Our results indicate that the inflammasome and the inflammatory reactions it triggers, play an important role in the emergence of tau pathology,” Michael Heneka, director of the Department of Neurodegenerative Diseases and Gerontopsychiatry at the University of Bonn and a senior researcher at the DZNE, said in a news release.
“It appears that inflammatory processes mediated by the inflammasome are of central importance for most, if not all, neurodegenerative diseases with tau pathology,” he continued.
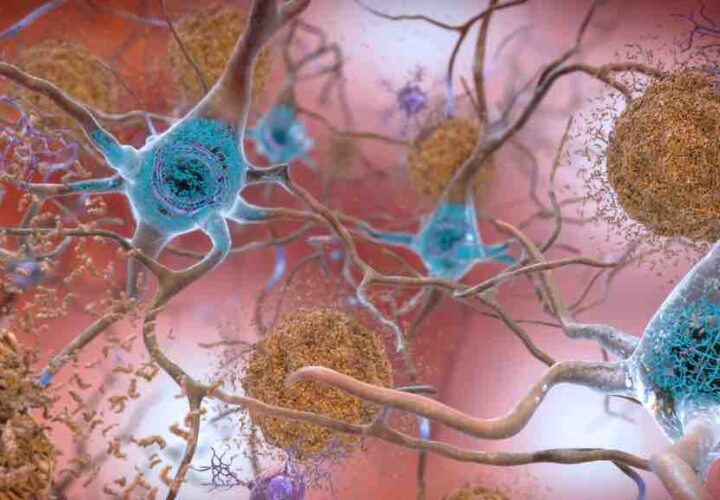
The study found that this inflammasome influences enzymes to cause a chemical change in tau proteins. In patients with Alzheimer’s disease in particular, the inflammasome causes amyloid beta (Abeta) molecules to accumulate in the brain in between neurons, a process that begins in the early stages of the disease before the destabilization of tau proteins.
The team had previously studied this accumulation of Abeta molecules, but this study was able to tie their previous research to the latest focus on tau proteins and inflammation.
“Our results support the amyloid cascade hypothesis for the development of Alzheimer’s. According to this hypothesis, deposits of Abeta ultimately lead to the development of tau pathology and thus to cell death,” said Heneka.
These findings are significant in demonstrating and better understanding the relationship between inflammation and FTD diseases. Neuroscientists see opportunities for therapeutic application of this new knowledge, including potentially containing tau pathology through medication that halts the immune response and thus inflammation.

Hi. I think I am on the verge of developing Alzheimers as I have become forgetful – to the point where I have to search through the cupboards to find items in the kitchen that have been in the same place for years. I also have a near constant head ache. My family and work colleagues have started to notice and it is becoming a real fear for me. I take asprin daily and have stopped drinking alcohol, a little late really as I have always enjoyed a drink (many times) more than was good for me). I recently had a brain scan that showed very minimal deterioration – in line with my age (70), but which was a relief as the consultant said it is an acceptable level and nothing to be concerned about. I would be happy to try any new drugs as I have seen in my work as a carer the devastation that this diseaze can bring.
My husband had a stroke from a bleed in the brain.The doctors did MRI and pathology,which showed it was from amyloid buildup.Hes in the hospital, and is been fed mostly carbs and sugars.Do you think they should put him on a no carb diet,based on the studies that this could make it worse?