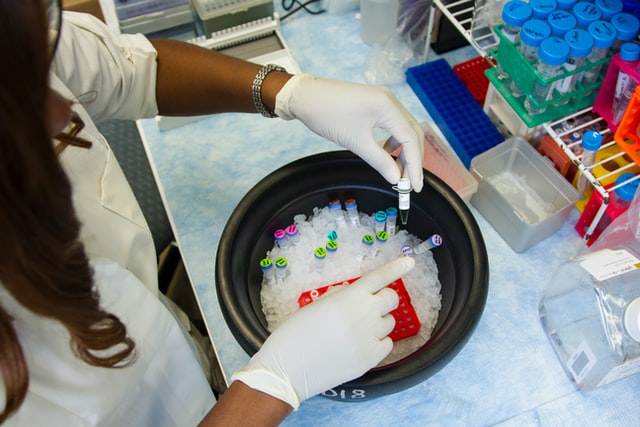
Longeveron’s investigational early-stage Alzheimer’s drug Lomecel-B, made from donated bone marrow, is enrolling participants for a clinical trial.
Scientists have faced obstacle after obstacle in the search for effective Alzheimer’s drugs. While there are a number of symptomatic drugs approved by the Food and Drug Administration for treating Alzheimer’s, none of them address the underlying disease. The safety and efficacy of the only FDA-approved anti-amyloid therapy on the market remain in question.
But, there is reason for optimism. Researchers and drug developers have been diversifying their approaches, exploring biological targets beyond Alzheimer’s mainstay, amyloid. Case in point: A cell therapy known as Lomecel-B is currently under investigation in a Phase 2a clinical trial.
The therapy’s developer, Longeveron, suggests that Lomecel-B can help reduce brain inflammation, improve the brain’s vascular system and promote the formation of new neurons. Lomecel-B is derived from special living cells known as Medicinal Signaling Cells (MSCs) that are derived from bone marrow tissue donated by healthy adults aged 18 to 45.
Scientists have identified amyloid plaques and tau as key biomarkers of Alzheimer’s, but Chris Min, Longeveron’s interim CEO said given the uncertainties surrounding the therapeutic benefits of targeting these pathologies, it is reasonable to explore alternative approaches to treating Alzheimer’s.
“One thing that I think is clear is that part of the damage to the brain that occurs in the process of Alzheimer’s disease seems to be due to inflammatory processes among other things. So Lomecel-B is an approach to addressing that part of the disease. I am not saying that one shouldn’t invest at first in those other parts of disease,” Min said of amyloid and tau. “But at the end of the day, it’s not clear that they will necessarily result in therapeutic benefits to patients.”
Currently, Lomecel-B, administered for Alzheimer’s via peripheral intravenous infusion, is in the early stages of clinical research. A Phase 1 trial, which involved 33 study participants with mild Alzheimer’s, reported that Lomecel-B infusions were safe and well-tolerated. Participants who received low-dose Lomecel-B experienced significant improvements in cognition, compared to the placebo group.
However, Min said it’s too early to bank on the results of this small trial. Further research is needed to confirm the finding, which ultimately may or may not hold up, he said. As of January, the Phase 2a clinical trial was seeking to enroll 45 patients to build on these findings, looking at single and multiple doses. (For more information, visit the trial page at clinicaltrials.gov; to reach out to the team behind the trial, click on Contacts and Locations in the green box at right.)
As Min reflects more broadly on the state of Alzheimer’s research, he urges those living with the disease to hold out optimism and to maintain their support for the research community.
“I just want to make sure patients know that there’s so many of us who are trying our best to come up with therapies for them,” Min said. “There have been some challenges across the space of therapeutics, and I just ask that they keep up their hope, and that they continue to support the NIH, that they support research in general. That’s really the path forward [with health conditions] like Alzheimer’s disease where there’s such a vast unmet medical need.”



I am in 5 clinical trials for Alzheimer’s through Stanford and I have to fly or drive myself and caregiver to the trials or do them through Zoom. The airline trials take a lot of my money and I usually have to pay for a hotel too. I will be giving to ALZ through the walk, and I just had to get a hot water heater and now my electricity seems to be in need of a new wiring, so I can’t afford to pay more now.. I already had to give up my long term care policy because I couldn’t cover the cost of the premium. I’m 77 (will be 78 in November) and am on a fixed income. I have mild cognitive impairment, not Alzheimer’s
I read that early trails are ongoing for anti-inflammatory agents for TBI. So more research is ongoing in this area. I’m an investigator on TBI. I can send you articles I’ve run across on the evolving literature of on neuroinflamation and TBI. Some promising new directions that might apply to Alzheimer.