Was it too good to be true? China last week awarded conditional approval for a new seaweed-based drug for Alzheimer’s, but experts in the United States are raising strong concerns.
The Alzheimer’s Association issued a statement from Maria Carrillo, chief science officer, saying that it “cannot recommend that anyone in the U.S. take this drug until it has met the safety and efficacy standards of the U.S. FDA.”
The drug, Oligomannate, which is distributed by Shanghai-based Green Valley Pharmaceuticals, uses an extract from brown algae. The company said it showed promise in slowing mild to moderate Alzheimer’s disease.
In a news release, the company said it would launch its drug “very soon” in China and said it planned to start a global phase 3 clinical trial early next year with sites in the United States, Europe and Asia.
Green Valley said that a trial in China involved 818 patients and showed results in as little as four weeks. But experts were wary.
Aaron Ritter, director of clinical trials at the Cleveland Clinic’s Lou Ruvo Center for Brain Health in Las Vegas, urged caution. “This is certainly not the panacea for Alzheimer’s disease that people are reaching for,” he said.
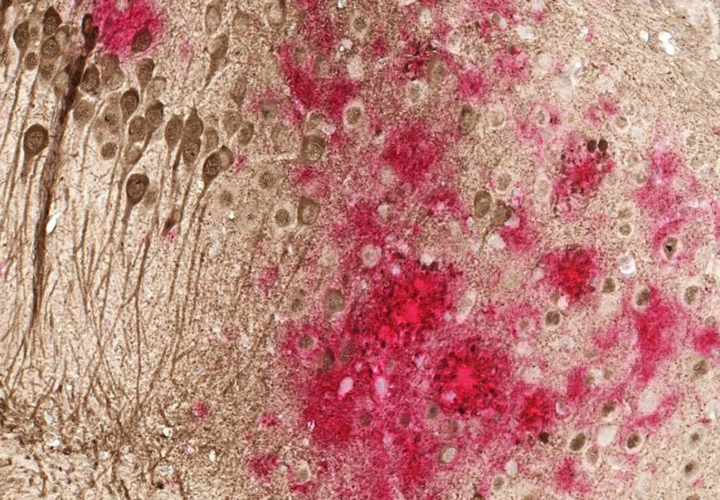
Last week’s news from China seemed to build on research into the connection between dementia, inflammation and bacteria in our digestive systems. Scientists are increasingly wondering whether inflammation in other body parts like the digestive system might have something to do with Alzheimer’s.
But the research is in its earliest stages. The Alzheimer’s Association said it is “intrigued” by the gut-brain and immune system thesis and Green Valley’s claims that its drug would benefit cognitive function, reduce amyloid buildup in the brain and protect other crucial tau proteins.
“That said,” the Association’s Carrillo said, “little is known at this point about exactly how the drug works.”
Perhaps the strongest cautions came from Derek Lowe, a scientist who writes In the Pipeline, a blog for the American Association for the Advancement of Science.
While noting that new approaches to Alzheimer’s are very much needed, Lowe initially wrote that “the improvements in this trial seem to come on very quickly, which makes me wonder, frankly, if they’re real and reproducible.”
He followed up that original caution with an even more skeptical take: “Overall, I am even less thrilled than I was yesterday about the prospects for this drug.”
“A new Alzheimer’s therapy is already going to face a tough development path and a lot of skepticism, but this one has even more red flags that I had realized,” he wrote. “More on this as it develops. But it is not developing well.”




good news.Wil wait and see