Resetting the brain’s immune cells and delaying chronic inflammation may help improve traumatic brain injury recovery, according to a new study.
Resetting the brain’s immune cells and delaying chronic inflammation may help improve traumatic brain injury recovery, according to a new study. Researchers at Trinity College Dublin and the University of Maryland School of Medicine tested this method in preclinical trials in mice. They found that targeting microglia, the brain’s immune cells, could be a promising preventive treatment for traumatic brain injuries and associated dementia.
Traumatic brain injuries (TBIs) like concussions were once brushed off as relatively insignificant events that someone could recover from in a few weeks. But as more research has linked TBI to neurodegenerative diseases like dementia and chronic traumatic encephalopathy (CTE), the stakes are higher when treating hits to the head.
In fact, research has shown that even one traumatic brain injury could double the risk of developing dementia later on.
Microglia’s Role in Neurodegeneration
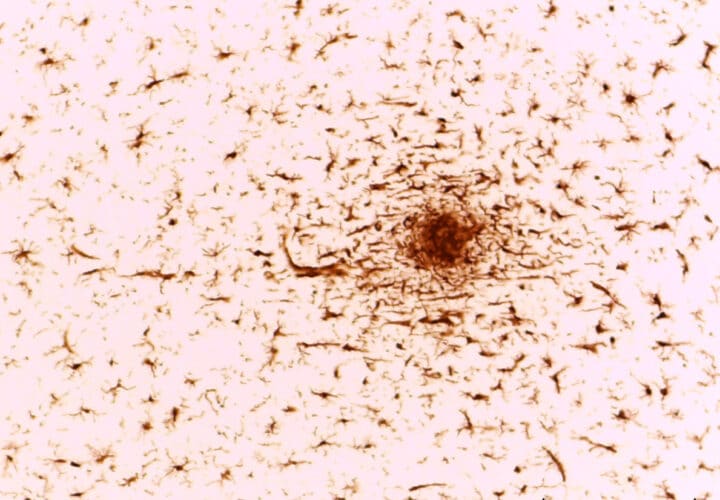
The study focused primarily on microglia, immune cells found in the brain, and their relationship to neuroinflammation.
In many ways, inflammation works as a good force that helps clean the brain. Microglia scavenge and clear damaging elements like toxic proteins from the brain and release cytokines, pro-inflammatory molecules. In Alzheimer’s, dementia and TBI, however, this inflammatory response is over-activated and can become harmful.
“Inflammation is normally a ‘good guy.’ It clears infections and helps heal wounds, for example,” Linda Van Eldik, director of the Sanders-Brown Center on Aging at the University of Kentucky, told Being Patient in a past interview. “But in Alzheimer’s disease, the inflammation somehow gets out of whack. It gets too strong and sustains for too long—it’s now turned into a ‘bad guy,’ destroying the neurons that carry signals from one part of the brain to the next.”
Microglia can also be triggered by traumatic hits to the head, releasing an inflammatory response in an attempt to protect to brain. But long-term inflammation leads to neurodegeneration and cognitive decline, such as in CTE, which has a higher risk of occurring among football players and other contact sports athletes.
Delaying Inflammation Leads to “Reset” of Microglia
In the new study, the researchers decided to try to delay this inflammatory response after a TBI to see if it had any therapeutic effect on the brain. In mice who had experienced a TBI a month earlier, the researchers inhibited a receptor in microglia that destroyed 95 percent of the immune cells.
This inhibition, or stopping of the microglia for about a week, “reset” the immune cells. The microglia eventually recovered after the halt. The researchers found the reset led to a lower inflammatory state, as well as fewer neuron deaths, less brain damage and improved cognitive performance in mice.
“These preclinical studies suggest that the consequences of TBI on brain degeneration and related neurological impairment may be modifiable quite a long time after injury by targeting inflammation pathways, which is a finding at odds with widely accepted views about head injury,” David Loane, research assistant professor at Trinity’s School of Biochemistry and Immunology, said in a news release.
Focusing on inflammation in the brain has been a growing area of interest among Alzheimer’s researchers who are searching for therapies beyond amyloid drugs.
Microglia in particular have been the focus of several recent studies on neurodegenerative diseases. In one study, researchers altered microglia in the brain to improve performance on memory tests by up to 50 percent.
“The exciting thing is the possibility that we may one day be able to minimize brain degeneration and neurological impairment in people who have suffered a TBI,” Loane said. “It will of course always be incredibly important to act quickly whenever someone suffers a TBI, but our findings suggest targeting inflammation pathways further down the treatment line may make a major difference to long-term brain health and recovery.”