AmyriAD Therapeutics CEO Sharon L. Rogers shares the scoop on AmyriAD's investigational Alzheimer's drug AD101.
The majority of experimental Alzheimer’s drugs are being designed to halt the progression of the disease — a strategy which so far has yielded a 99-percent failure rate. Trials for these drugs — mostly expensive antibody-infusion based therapies — focus on treating people with Alzheimer’s early on. However, there are currently six million Americans already living with Alzheimer’s, and the majority are too far along in their Alzheimer’s journey to qualify for the trials.
AmyriAD is a pharmaceutical company led by CEO Sharon L. Rogers. While working at Eisai, Rogers lead the development of the cholinesterase inhibitor donepezil (Aricept). Now, she and her team at AmyriAD are focusing on complementing existing treatments to improve cognition and quality of life through a new therapeutic mechanism.
In November of 2021, the company announced that their new symptom-managing drug, AD101, would enter Phase 3 clinical trials by the end of the year with hopes that it could also alleviate some of the burden faced by caregivers.
In a November 29, 2022 follow-up, the company announced: “The team is now gearing up to launch our Phase 3 clinical trial program in the first half of 2023. These trials will include patients across a broad range of disease severity reflecting real-life clinical practice.”
There is a great deal of discussion in the drug development world about finding a cure to Alzheimer’s, the prevalence has reached epidemic proportions around the globe. Rogers, however, isn’t only interested in a cure: She said AmyriAD is focused on improving quality of life for Alzheimer’s patients and their families: creating better pharmaceutical options for symptom management.
“You could have the world’s best drug for modifying the course of the disease,” Rogers told Being Patient. “But the fact of the matter is most patients do not get to a care provider to be diagnosed until they’re at the early moderate stages at best.”
An accessible treatment for later stages of Alzheimer’s disease
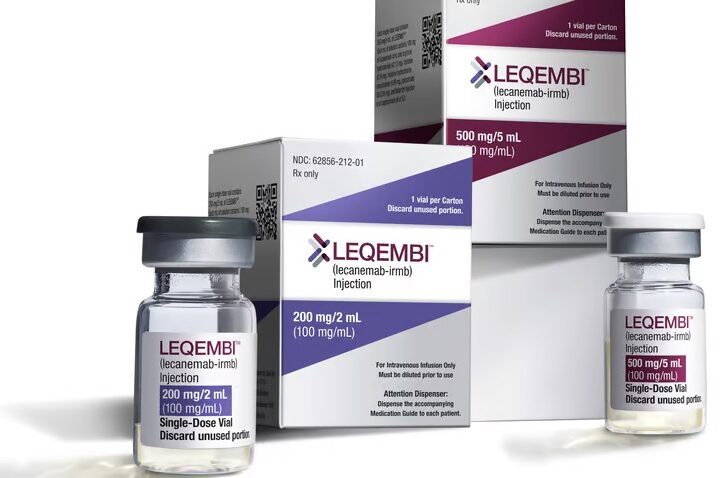
While anti-amyloid based drugs like lecanemab are promising candidates for treating Alzheimer’s, they are tested in a very small subset of the population: people with mild cognitive impairment, or, people who have biomarkers of Alzheimer’s detected so early on, symptoms have not yet even appeared.
The only way currently available to detect these biomarkers before symptoms develop is a lumbar puncture or a PET scan (which can cost thousands of dollars if not covered by insurance). In order to be eligible for approved anti-amyloid drugs like Aduhelm, patients are on the hook to pay for PET scans which aren’t currently covered by Medicare for the diagnosis of Alzheimer’s.
(In February 2024, Biogen took Aduhelm off the market indefinitely.)
Lecanemab and similar anti-amyloid drugs that target beta-amyloid only work to reduce the levels of toxic beta-amyloid. “You have to do more than that,” Rogers said. “[They] don’t manage their symptoms by any kind of direct manner, management or mechanism.”
AmyriAD’s AD101 is designed to complement the effects of the cholinesterase inhibitor donepezil (Aricept).In the company’s Phase 2 trials, AD101 improved both cognition and global function. Building off these results, the Phase 3 trial may set the stage for a broad application for the drug’s approval.
“We’re aiming for a broad label for Alzheimer’s disease so mild, moderate and severe,” Rogers said. “Contrary to popular belief, it is good to treat people as long as you possibly can, even when they’re in a nursing home, because there is still a benefit to therapies like donepezil and then hopefully AD101.”
A new therapeutic pathway
AD101 is designed to counteract Alzheimer’s cognitive symptoms by binding to proteins called T-type calcium channels found in the hippocampus and in neurons that release acetylcholine. These proteins are like gates that open in response to a specific electrical signal, controlling the flow of calcium within the neuron. When AD101 binds and modifies the activity of this protein, it pumps out more acetylcholine. T-type calcium channel proteins are also found on lysosomes — cellular recycling factories — which could also regulate levels of a key Alzheimer’s biomarker, beta-amyloid plaques, and contribute to overall brain health.
If AD101’s Phase 3 trial is successful, it could usher in more symptom-treating drugs for dementia that target calcium channels and lysosomes.
Seeking a broad label that encompasses people at all stages of the disease, Rogers and AmyriAD hope that this new Alzheimer’s drug could benefit millions of people with Alzheimer’s and their caregivers worldwide. The Phase 2 trial found that complementing donepezil (Aricpet) with AD101 doubled the amount of patients who improved their global function. Outcomes will be different for every patient and caregiver, but Rogers’s hope is that it will help people maintain their autonomy for longer.
“Maybe it’s making a sandwich, maybe it’s going to the grocery store, maybe it’s being able to select their clothes and dress themselves,” Rogers said. “That’s pretty meaningful for day to day function, and easing the amount of time that [a caregiver] has to devote to that loved ones or patients.”
CORRECTION, 14 December, 2022 at 11:34 A.M. ET: Rogers’ name was updated from Sharon Rogers to Sharon L. Rogers to avoid confusion with Captain America.
UPDATE, 14 December, 2022 at 11:37 A.M. ET: An AD101 Phase 3 trials timeline update was added from a November 29 press release.
UPDATE: 3 March 2024, 9:18 P.M. ET. In February 2024, Biogen took Aduhelm off the market, citing financial concerns. Although the drug did receive accelerated, conditional FDA approval for the treatment of early Alzheimer’s disease in 2021, it is no longer available to new patients. The company announced it would sunset trials in May 2024 and cease supplying the drug to current patients in November 2024.


We are in our 7th year of dealing with Alzheimer’s. My wife was in Roche’s trial for ganaterumumab but they stopped the study. I’m glad to hear that it’s reached breakthrough status.